How to Prevent Thermal Runaway in a Lithium-ion Battery?
Boeing’s Dreamliner 787, which Boeing advertised as 20% fuel efficient, was grounded in 2013. In the same year, Tesla’s Model S came under a federal safety investigation after it caught fire at least 3 times. In 2016, Samsung recalled 2.5 million Galaxy Note 7 smartphones. For all three companies, which are top players in their domain, the problem was the same – thermal runaway in the Lithium-Ion battery installed in the heart of their product as a power source.
Fast forwarding to today, battery-related explosions have increased five times since 2016.
Even though lithium-ion batteries are technically safer and less likely to fail today than a decade ago, companies haven’t been able to completely eliminate the issue. And the use of lithium-ion batteries has increased so much more in numbers. With so many battery-operated appliances, from drones to automobiles, companies are still searching for a solution. One such company approached us with the same. Their problem statement – how to stop lithium-ion batteries explosion and are there some solutions that can be adopted?
How do lithium-ion batteries catch fire and explode unexpectedly?
Disclaimer: We have concealed some details due to confidentiality reasons.
Li-ion batteries are the most used type of batteries across several industries especially electronic appliances but are also an unattractive solution for others. Do you know what makes them hazardous?
As the title suggests, one of the major reasons why most lithium-ion batteries explode is because of thermal runaway. But, what exactly is thermal runaway?
At its core, thermal runaway is the dispersion of the excess heat in lithium-ion batteries. The overheating causes a chemical reaction that results in fire or explosion.
So how to prevent thermal runaway in a lithium-ion battery?
Our search team did some digging and returned with a set of available solutions. R&D teams can evaluate and adopt one or more of these solutions for quick implementation. While some of the most promising solutions were provided to the client, below are the three exemplary solutions companies can adopt.
Is your team also looking for solutions to prevent thermal runaways? We can find ’em for you.
1. Introducing a Flame Retardant
Thermal runaway often occurs from punctures and improper charging. To counter such fire hazards, the inventors used a thermal fluid that contains a flame retardant.
A flame retardant is a compound that inhibits, suppresses, or delays the production of flames or prevents the spread of fire.
Here they have microencapsulated the flame retardant (usually a bromine compound) in high-density polyethylene and added water and a glycol compound to prepare the thermal fluid used. The glycol compound is used here as “antifreeze” (common glycol compounds used are ethylene glycol, diethylene glycol, and propylene glycol). 3M Innovative Properties Co. was recently awarded a patent (WO2022118246A1) for the elements and the process of an encapsulated fire retardant.
Also, the invention is mostly discussed in light of EV batteries. A battery when called upon to power an electric vehicle heats up. Thermal fluid flows through the container and over the modules of the battery. Porche AG, Volkswagen, and Audi AG even filed a patent (US10967210B2) on the same.
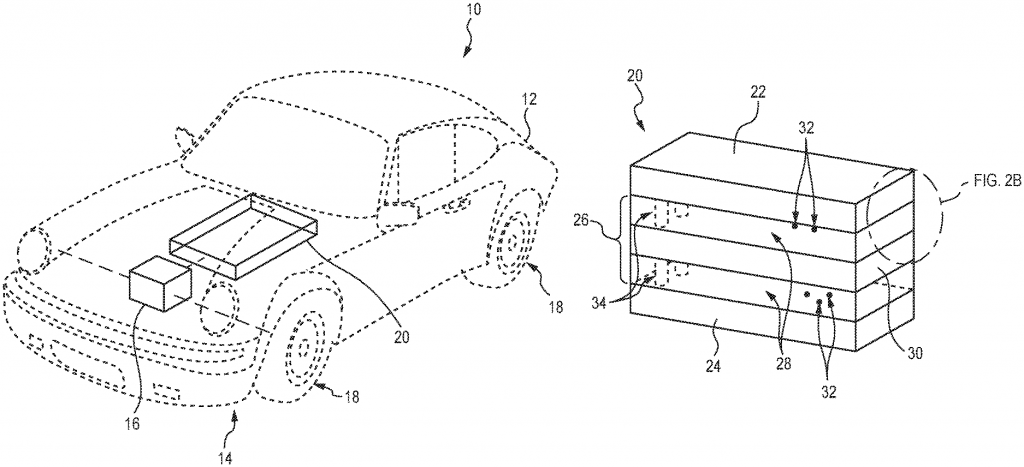
In the event of an overcharge, or a car accident resulting in a battery puncture, the flame retardant in the thermal fluid acts to reduce the fire hazard. More precisely, because of excess heat of the fire, bromine compound microcapsules rupture on reaching the rupture temperature. The flame retardant is released from the microcapsules and acts to bring the fire under control.
2. Using Damage initiating Devices
The University of California researched a solution related to high elastic modulus polymer electrolytes suitable for preventing thermal runaway in a lithium-ion battery. Also, they have developed a thermal runaway shutdown mechanism that can be triggered either mechanically or thermally (or both), as battery damage happens (i.e., before or shortly after thermal runaway starts), and take care of the problem before it can even begin. Their patent US20160126535A1 discusses the same.
Such predictive or instantaneous countermeasures are especially needed when a battery is subjected to impact or high pressure and its internal structure gets damaged, causing internal shorting.
Damage initiators can trigger widespread damage or destruction of the electrode as a mechanical load is applied to the battery. Due to this, the internal resistance increases significantly to mitigate thermal runaway even before it can happen.
Here they have talked about two types of damage initiators –
Passive damage initiators
These initiators initiate cracking or voiding in electrodes upon impact. Such cracks and/or voids increase the internal impedance of the electrode reducing the heat generation associated with possible internal shorting. Such additives are known as cracks or voids initiators (CVIs).
The debonding or stiffness mismatch of CVI-electrode interfaces, fracture, rupture of CVI, etc. causes the electrode damage. Examples of passive additives include solid or porous particles, solid or hollow/porous fibers, tubes, etc. Carbon materials such as graphite, carbon nanotubes, activated carbons, carbon blacks, etc are used to form these additives.
Active damage initiator
These initiators can produce a significant volume or shape change upon mechanical or thermal loading. Active damage initiators can include solid or porous particles, solid or hollow beads, solid or hollow/porous fibers, tubes, etc. Shape-memory alloys such as Ni—Ti, Ni—Ti—Pd, Ni—Ti—Pt, etc. form the active damage initiators.
3. Changing the Battery Design for Minimal Impact
According to a patent filed by Tesla, the inventors discussed a battery design that provides a predictable pathway through a portion of the cell (e.g., the cell cap assembly) for the efficient release of the thermal energy that occurs during thermal runaway, thereby reducing the chances of a rupture in an undesirable location.
Furthermore, the design maintains the functionality of the cell cap as the positive terminal of the cell, thereby having minimal impact on the manufacturability and maximum use in a variety of applications.

While searching for solutions to prevent thermal runaway in a lithium-ion battery, we also found some quality solutions to other problems in the LIB domain. Problems such as-
- How to overcome dendrite formation?
- How to increase the life cycle of Lithium-ion batteries?
- How to decrease viscosity and increase the dispersion efficiency of anode binders?
- How to you can make your LIB operate at high temperatures?
Seeking solutions for the above-mentioned problems? We might be able to put an end to your woes.

-
[…] data at the cell level. Monitoring each cell can help with early warnings for potential hazards, such as thermal runway. Lithium batteries are also prone thermal runway, but that is the main reason all lithium ion […]
[…] Preventing thermal runaway initiates with meticulous battery design, incorporating features that prioritize safety. […]
Leave a Comment






Good write-up. Thanks. The concept of Active Damage Initiators is not clear from the above. Request more information.
Limiting charge rates is also a way to mitigate the risk. Also narrowing the Ah discharge window reduces thermal build-up.
The battery management system ensures that the battery remains in its specified working range. This requires extremely precise measurement of the charge and discharge currents, cell voltage, and temperature. Great info you have shared here.