As a litigator, you love killer prior art, don’t you? A perfect Tier-I reference(s) would make life so easy and court proceedings bliss. But what if killer prior art does not exist?
The next possible solution would be to prove ‘lack of inventive step’ based on obviousness. In pharmaceutical patents especially, obviousness is a hot topic, partly due to the structural similarity among completely different active pharmaceutical ingredients. Even a small change in the structure could entirely change the properties of the compound.
So, how can one prove obviousness in pharmaceutical patents?
Well, you can argue that the invention is “obvious to try”. You see, there are a finite number of possible solutions to a problem, and if you can prove that a person skilled in the art could reach that solution suggested in the patent, you can prove the patent invalid based on obviousness.
Easy peasy, lemon squeezy? Well, kind of. We try to challenge the validity of pharma patents a lot on the grounds of obviousness.
Want to know how we approach it? Let me take you through one such recent case where we proved the patent invalid.
Our Client Mark wanted us to do a litigation grade search to prove a patent invalid
In this case, the patent in question claimed the usage of mass spectrometry to determine the quantity of ‘vitamin D’ in a blood sample. Before going through the quantification process, vitamin D should be modified with 4′-carboxyphenyl-TAD(main claim element). The objective behind this concept is, sample modification enhances ionization and helps in measuring analytes even in a small concentration as well.
Finding this exact structure was going to be very challenging. Keeping this in mind, we started our hunt with the structure search on PatSnap, STNext & SciFinder to check every 4′-carboxyphenyl-TAD derivative i.e. low hanging fruits. But to no avail.
We next analyzed the theses, research abstracts, publications, symposia, seminars, etc. of universities having active contributions in this domain.
While scrutinizing the universities documents, we got a hint that Asian institutes are more into this direction, so we specifically explored data of Asian countries such as China, Japan, Korea at their official databases like – National Institute of Japanese Literature, KIPRIS, J-PLATPAT, Asian Reading Room, CiNii, J stage, etc.
From here, we got our hands on prior art that indicated modification of vitamin D with phenyl-TAD (PTAD) instead of the desired 4′-carboxyphenyl-TAD derivatization.
The below illustration shows the difference between what we needed vs. what we got –
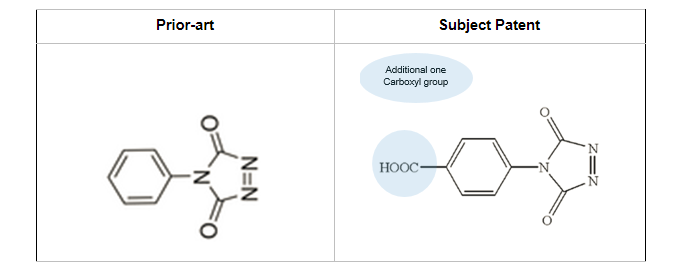
Looking at the picture, you’d agree that we were back to square one.
However, we did not lose hope and went deep into the chemistry behind this modification reaction (i.e. vital part of invention – use of 4′-carboxyphenyl-TAD to bind with vitamin D).
It is essentially Diels-Alder Reaction at work where a diene (vitamin D) and Dienophile (4′-carboxyphenyl-TAD) are joining with each other. Anyone with a basic knowledge of chemistry would know for a fact that the presence of an electron-withdrawing group (e.g. carboxy) on any dienophile (e.g. PTAD) would enhance its activity.
So, isn’t it ‘obvious to try’ 4-carboxy-PTAD instead of PTAD?
As we know, we can’t simply prove obviousness in pharmaceutical patents. So, we backed our case by couple of more findings –
- People in that era were trying to make derivatives of vitamin D with other Cookson-type reagents, including MBOTAD, DMEQTAD, NPTAD as well. (e.g. Derivatization Of Neutral Steroids)
- Also, we showcased as a reference one US publication (US6399796B2) which talks about modification of steroids (vitamin D analogue) with 4′-carboxyphenyl-TAD.
Both of these findings indicate that one can think of trying different modification agents and would end up using 4′-carboxyphenyl-TAD.
Concluding Notes
In our years of conducting prior art searches, we have figured that there are always intelligent ways to approach a search. You see, sometimes, a seemingly random piece of art has more to tell than one can expect at first glance.
You just need to have a keen eye for detail and grit to go on amidst the tough waves. Prior art searches can range in difficulty from a bit to a whole new level of complicated. However, we found in about 8 of these 10 cases that prior art can be found, with a combination of grit and investigative approach.
Below are few cases where above mentioned strategies can be helpful:
- Novartis Pharmaceuticals Corporation v. Lupin Inc. 1:21-cv-01105
- 1-21-cv-01107 (DDE) Novartis Pharmaceuticals Corporation et al v. Lotus Pharmaceutical Co., Ltd. et al
- 1-21-cv-00992 (DDE) Astellas Pharma Inc. et al v. Alkem Laboratories Ltd.
- Novartis Pharmaceuticals Corporation et al v. Dr. Reddys Laboratories, Inc. et al 1:21-cv-01106
In this case too, digging deep behind the chemistry of the claim element helped us prove the invalidity of the patent, and turned a 103 reference into outstanding 102 prior art. Want to know more about our investigative approach to patent searches? Click here to read everything about it.
Authored by Divya Goyal, Lead, Pharma team